Tuning metal nanoparticles on a support
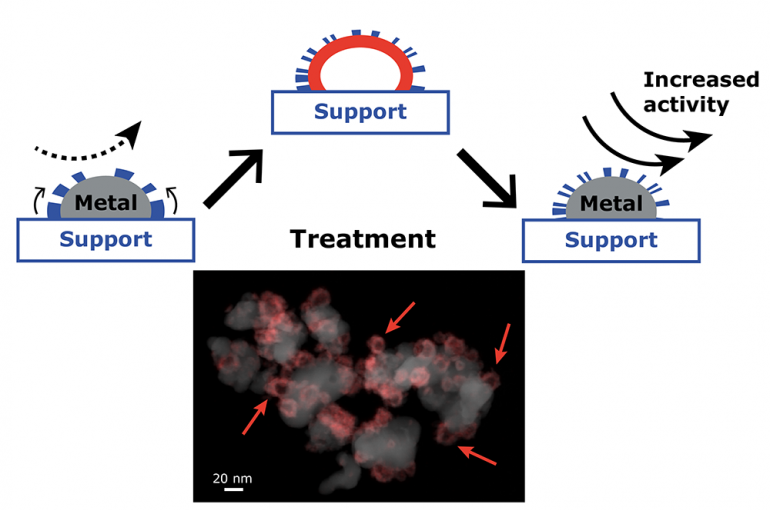
Metal nanoparticles are the ‘workhorses’ in many catalysts that speed up and steer major chemical reactions. They are widely used in the chemical industry to produce fuels and chemicals such as plastics, pharmaceuticals and fertilizers. To space the nanoparticles evenly and hold them in place throughout the reaction, the nanoparticles are distributed over a support material. Support materials may decorate the surface of the nanoparticles, just like chocolate sprinkles over ice-cream, and bring about new beneficial properties to the nanoparticle. However, too many sprinkles make it impossible to taste the ice-cream, just as in our case, where extensive coverage of the nanoparticles by the support decreases their activity. Ideally, we would like to have enough of this decoration over the metal nanoparticle, a bit of sprinkles, but not too much that the nanoparticle ends up completely covered and inactive.
Researchers Carlos Hernández Mejía, Tom van Deelen and Krijn de Jong found a facile method to effectively control this decoration over the nanoparticles that involves expansion of the nanoparticles followed by shrinkage to their original size. This optimizes the coverage of the nanoparticles by the support thus leading to enhanced activity of our nanoparticles. This method to tune metal-support interactions is valuable tool for designing catalytic materials with improved performance. The results are published today in Nature Communications.
Publication
Activity enhancement of cobalt catalysts by tuning metal-support interactions
Carlos Hernández Mejía, Tom W. van Deelen & Krijn P. de Jong
Nature Communications 9, 4459 (2018), DOI 10.1038/s41467-018-06903-w

