Researchers show how an antidepressant drug can inhibit viral infection
Researchers have shown how the antidepressant drug fluoxetine can bind to and inhibit a key protein in the replication machinery of enteroviruses. These viruses infect millions of people yearly, and can cause severe and occasionally life-threatening diseases. The discovery could pave the way for development of antivirals against these important human pathogens. The research was done at Utrecht University in collaboration with Aix-Marseille University and the results were published in the journal Science Advances on the 5th of January.
Viruses pose a tremendous threat to humanity, as painfully demonstrated by the COVID-19 pandemic. Antiviral drugs can bind to viral proteins and inhibit their function, thus preventing virus replication and reducing disease symptoms. However, it is expensive and time-consuming to develop such molecules from scratch, and to assess their safety and effectiveness in clinical trials.
Teaching old drugs new tricks
An alternative strategy to identify antiviral drugs is to screen existing libraries of FDA-approved drug molecules to see if they inhibit a particular virus. The advantage of this is that these molecules have already been through clinical trials and are proven to be safe in humans. However, “finding a drug that inhibits a virus is only the first step”, explains co-lead author Daniel Hurdiss. “Figuring out exactly how that drug inhibits a virus is the hard part.”
A detailed understanding of how fluoxetine can inhibit replication was unknown until now.
By understanding how a small molecule inhibits a virus, researchers can use this information to develop more potent molecules with a higher barrier to antiviral resistance. Previous studies by the Virology group in Utrecht identified several FDA-approved molecules, including fluoxetine, that can inhibit enteroviruses replication, but a detailed understanding of how this occurs was unknown until now.
Millions of infections and no antivirals
Enteroviruses are a large group of viruses that includes rhinoviruses, causing the common cold, coxsackieviruses, that are an important cause of viral meningitis and encephalitis, and the poliovirus, that causes paralytic poliomyelitis. Collectively, these viruses are responsible for millions of infections each year. There are currently no licensed antivirals to treat enterovirus-associated diseases.
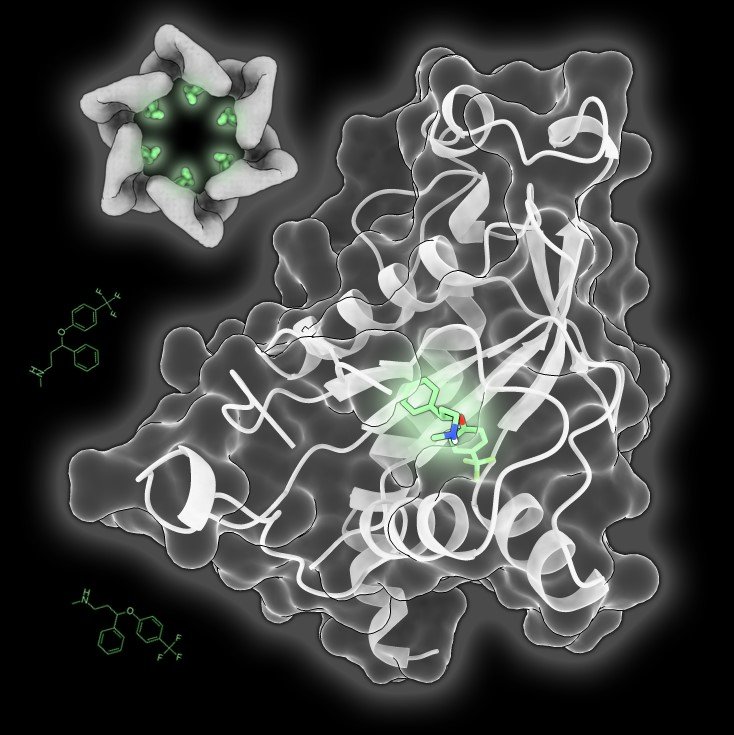
Throwing a spanner in the works
During an infection, these viruses hijack our cells and turn them into virus-producing factories. To achieve this, enteroviruses encode their own replication machinery, a crucial component of which is the 2C protein. In the paper by Hurdiss et al, researchers used a combination of structural biology and biochemistry techniques to show that fluoxetine binds to the 2C protein and prevent its function by locking it in a fixed conformation. “Just like a car engine, the components of the enteroviruses’ replication machinery must be able to move freely to work. By ‘stalling’ the 2C protein, fluoxetine inhibits efficient virus replication. These data provide the first detailed insights into how the enterovirus’ 2C protein can be inhibited by small molecules and should facilitate further development of anti-enteroviral drugs to mitigate the impact of these viruses on society.”
Daniel Hurdiss is an Assistant Professor in the Virology group of the Faculty of Veterinary Medicine, Utrecht University. He was recently awarded an NWO Veni grant to continue his enterovirus research.
Frank van Kuppeveld is Group Leader and Head of the Virology group of the Faculty of Veterinary Medicine, Utrecht University.

